What is Omenn Syndrome?
Omenn syndrome is a form of severe combined immunodeficiency [1,2,3]. Severe combined immunodeficiencies (SCIDs) are genetic disorders characterized by deficiencies in an individuals T cells and B cells [2]. What makes Omenn Syndrome unique is that, in addition to the deficiency in an person's immune cells, patients present with autoimmune symptoms [1,2,3]. It is characterized by T cells that do not need to recognize pathogens or invasive particle, to initiate their effects and IgE antibodies [1,3]. IgE antibodies are normally involved in fighting large pathogens, and are the antibodies responsible for allergies in humans. These all result in the characteristic symptoms of Omenn syndrome: chronic inflammation of the skin (a large red rash covering the body), swollen lymph nodes, swollen spleen, diarrhea, enlarged liver, and an increased risk for infection [1].
What are T cells and B cells?
|
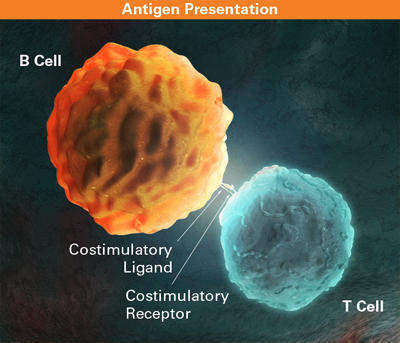
The immune system is broken up into two parts: the innate immune system and the adaptive immune system. The innate immune system is a fast responding, constant reaction to pathogens. The cells involved recognize common shared patterns found in pathogens. They react the exact same way to every pathogen that they enounter. The adaptive immune system is a slower, but more specialized portion of the immune system. They have receptors that detect highly specific parts of proteins found in pathogens, or epitopes. When they encounter these epitopes, the cells become activated and start fighting the pathogen. The response to the pathogens are highly specialized. For B cells, they become antibody producing factories, secreting the B cell receptor in a modified form into the bloodstream. These antibodies will be secreted into the blood stream and bind to pathogens. This will either directly inactivate the invading particle in some way, or it will mark the cell for destruction by the innate immune system. T cells come in two different varieties: CD8+ T cells, or cytotoxic T cells, and CD4+ T cells, or T helper cells. T helper cells recognize different types of pathogens, like viruses, bacteria, and fungi, and release signaling molecules to direct the immune system to respond properly. Cytotoxic T cells can kill invading pathogens by either engulfing them through phagocytosis or killing them by releasing toxic molecules into the body. [4]
|
Figure 1: A cartoon of a B cell and a T cell interacting
|
What is Rag1 and how does it cause Omenn syndrome?
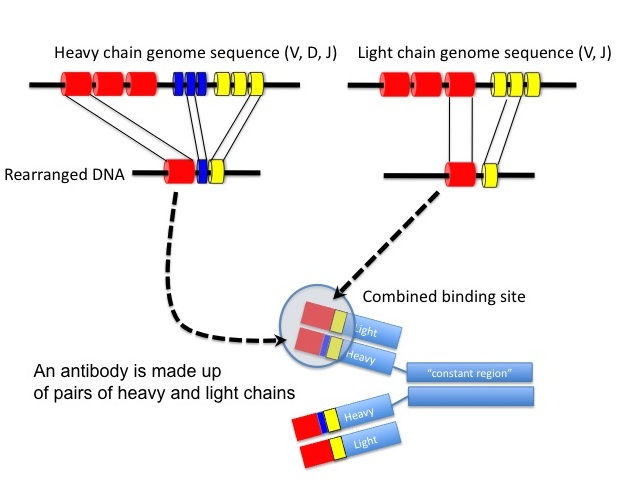
Humans are capable of creating 10,000,000 unique B cell receptors. The diversity found in T cell receptors is similar. In order to encode for each of these antibodies separately in the genome, it would require 2x10^10 base pairs of DNA. In contrast, the whole human genome is a little more than 3x10^9 base pairs total. In order to create the massive diversity of B cell and T cell receptors, organisms use a process cause "somatic recombination". [4] In this process, different pieces of DNA that are not adjacent in the genome are rearranged by RAG1 to create one functional gene product [4,5].
Figure 2: An illustration of somatic recombination in the process of forming an antibody. In the heavy chain of the antibody, segments from regions of the genome known as V (variabliity), D (diversity) and J (joining) are combined randomly to create a unique combination. Light chains do not contain the D region.
After an antibody is made, it is then tested in either the thymus or bone marrow to ensure that it is not self-reactive, called selection [4]. Individuals with Omenn syndrome have a mutation in the RAG1 gene that results in a protein with a greatly diminished ability to perform the somatic recombination necessary to produce T- and B-cell receptors [1,2,4,5]. This leads to the characteristic immunodeficiency found in the disease. The cause of the autoimmune symptoms has yet to be fully explained, but it is believed to be a problem in the selection of B cells [3].
Prognosis and Treatment
|
Without treatment, babies born with Omenn syndrome die rapidly, normally due to an opportunistic infection [1,6,7,8]. The only treatment available now is a full bone marrow transplant [6,7]. After a bone marrow transplant is done, patients require immunosuppression throughout their life to manage autoimmun symptoms. Research is being done into other possible treatments for Omenn syndrome, such as gene therapy [8].
|
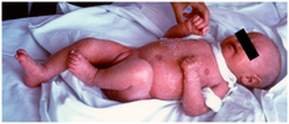
Figure 3: A baby with Omenn Syndrome. The skin is red and scaly due to an autoimmune reaction.
|
Why care?
Omenn syndrome is a relatively rare condition, only affecting 1 out of every 50,000 individuals [9]. Additionally, with a bone marrow transplant, an affected individual can lead a fairly normal life [6,7]. So the question remains, why should research be done into this rare, treatable condition? Studying Omenn syndrome could provide an interesting insight into how our immune system works. There remain many questions specific to Omenn syndrome that perplex scientists. Why do individuals with this disease produce such high levels of IgE? Why are the T cells that are produced self reactive? How do these cells arise in the first place? Understanding these questions could lead us to a more holistic understanding of the function of the immune system. This information could be used in coming up with new treatment for diseases like AIDS or autoimmune disease.
References
|
1) Marrella, V., Poliani, P.L., Sobacchi, C., Grassi, F., Villa, A. (2008). Of Omenn and mice. Trends in Immunology 29:133-140
2) Niehues, T., Perez-Becker, R., Schuetz, C. (2010). More than just SCID—The phenotypic range of combined immunodeficiencies associated with mutations in the recombinase activating genes (RAG) 1 and 2. Clinical Immunology. 135:183-192 3) Villa, A. (2011). Omenn Syndrome: inflammation and autoimmunity. J Transl med. 9(suppl2): 15 4) Owen, J., Punt, J., Stranford, S., Jones, P., Kuby, J. (2013). Kuby Immunology (7th ed). New York : W.H. Freeman and Company 5) Online Mendelian Inheritance in Man, OMIM (TM). Johns Hopkins University, Baltimore, MD. RAG1: {179615}. Last updated: {11/8/2014}. 6) Geha, Raif; Notarangelo, Luigi (2012). Case Studies in Immunology: A Clinical Companion (6th ed.). Garland Science. 7) Gomez, L., Le Deist, F., Blanche, S., Cavazzana-Calvo, M., Griscelli, C., Fischer, A. (1995) Treatment of Omenn Syndrome by bone marrow transplantation. The Journal of Pediatrics. 1:76-81 8) van Til NP, Sarwari R, Visser TP, Hauer J, Lagresle-Peyrou C, van der Velden G, et al. Recombination-activating gene 1 (Rag1)–deficient mice with severe combined immunodeficiency treated with lentiviral gene therapy demonstrate autoimmune Omenn-like syndrome. J Allergy Clin Immunol 2014;133: 1116-23. 9) Schwartz, R.A. (2014, Nov 7). Omenn Syndrome. Medscape. Retrieved 2/18/15 from http://emedicine.medscape.com/article/887687-overview#a0101 |
IMAGE REFERENCES
Banner image: http://beyondessential.com/healthy-immune-system/ Figure 1: http://mol-biol4masters.masters.grkraj.org/html/Cell_And_Molecular_Immunology5-T_And_B_Cell_Activation_And_Immunity.htm Figure 2: https://ittakes30.wordpress.com/2011/08/11/sequencing-the-immune-response/ Figure 3: http://en.wikipedia.org/wiki/Omenn_syndrome#mediaviewer/File:Omenn_syndrome.png |